The media partners found that, across all types of medical devices, more than 1.7 million injuries and nearly 83,000 deaths were reported to the FDA over the last decade. Reporters analyzed millions of medical records, recall notices and other safety warnings, in addition to interviewing doctors, patients, researchers and whistleblowers. 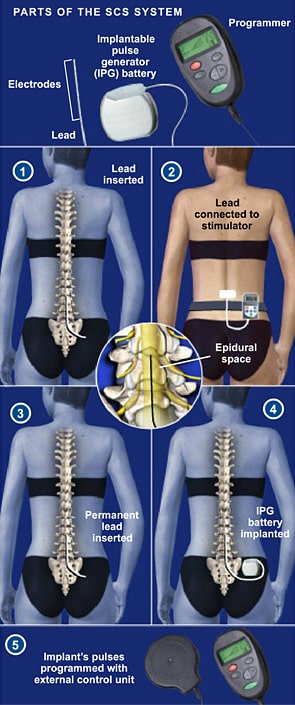
The AP reported on spinal stimulators as part of a nearly yearlong joint investigation of the global medical devices industry that included NBC News, the International Consortium of Investigative Journalists and more than 50 other media partners around the world. Most of these devices have been approved by the FDA with little clinical testing, however, and the agency's data shows that stimulators have a disproportionately higher number of injuries compared to hip implants, which are more plentiful. Medical device manufacturers insist spinal-cord stimulators are safe - some 60,000 are implanted annually - and doctors who specialize in these surgeries say they have helped reduce pain for many patients. Among the 4,000 types of devices tracked by the FDA, only metal hip replacements and insulin pumps have logged more injury reports. Patients report they have been shocked or burned or have suffered spinal-cord nerve damage ranging from muscle weakness to paraplegia, FDA data shows. They account for the third-highest number of medical device injury reports to the Food and Drug Administration, with more than 80,000 incidents flagged since 2008. and as a treatment for an aging population in need of pain relief.īut the stimulators - devices that use electrical currents to block pain signals before they reach the brain - are more dangerous than many patients know, an AP investigation found. Companies and doctors push them as a safe antidote to the opioid crisis in the U.S. "But look at me."įor years, medical device companies and doctors have touted spinal-cord stimulators as a panacea for millions of patients suffering from a wide range of pain disorders, making them one of the fastest-growing products in the $400 billion medical device industry. "I thought I would have a wonderful life," Taft said. Today, the 45-year-old Taft is virtually paralyzed, barely able to get to the bathroom by himself. After an operation to repair it, he said, the device shocked him so many times that he couldn't sleep and even fell down a flight of stairs. Being the only company with Level 1A evidence gives confidence in replicability of outcomes while reducing bias.Īs would be said on a television commercial - “come for the why of Eterna, but you’ll stay for the how.Taft's stimulator failed soon after it was surgically implanted. An unmatched breadth and depth of data in the spinal cord stimulation space. BurstDR uniquely modulates the medial pathway, thus addressing the suffering component of pain ( ).Ĥ. Patients do not seek out a pain physician due to painfulness, but rather due to the associated suffering resulting from their chronic pain.  
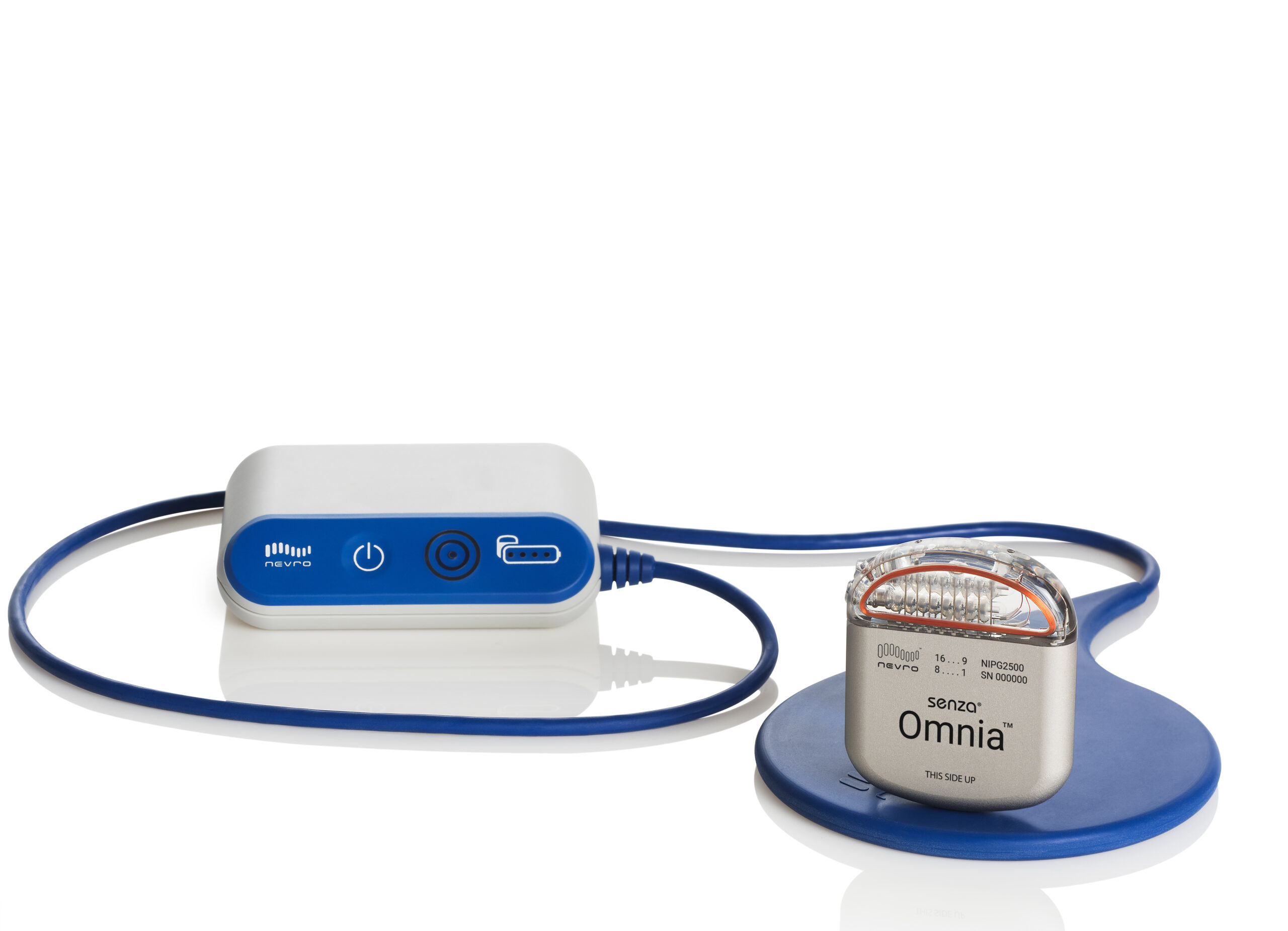
Where others are running constant current, we are uniquely able to cycle stimulation on and off ( ).ģ. Where others are using milliamps, we are using a fraction of that amplitude. This allows therapy to be delivered in a more efficient manner. BurstDR is the only low energy waveform on the market. Mimicking the body’s natural neural bursting creates stronger, more durable action potentials.Ģ. DeRidder’s patent on Burst stimulation (Patent No.: US 8,224,453 B2) results in Abbott being the only company not running tonic based stimulation. Apple devices with Bluetooth connectivity, an easy to navigate user interface, and a wireless charging puck does the trick here.ġ. Smaller incisions make approximation quicker, easier, and long term pocket site comfort more likely.ģ. Reduce the need for charging, you reduce the possibility for the aforementioned issues. All this increases frustration with systems. Additionally, issues accompany charging (components that fail, IPG’s that move, technical problems that arise, etc…). Patients do not want to spend significant amounts of their life charging a device. Feedback form patients and providers told us what they desire in a rechargeable battery. Now that we/ Abbott have officially launched #Eterna, I think it’s time to reflect on why and how this advancement is possible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
